Olympus Fluoview FV1000
Room 214.00.13
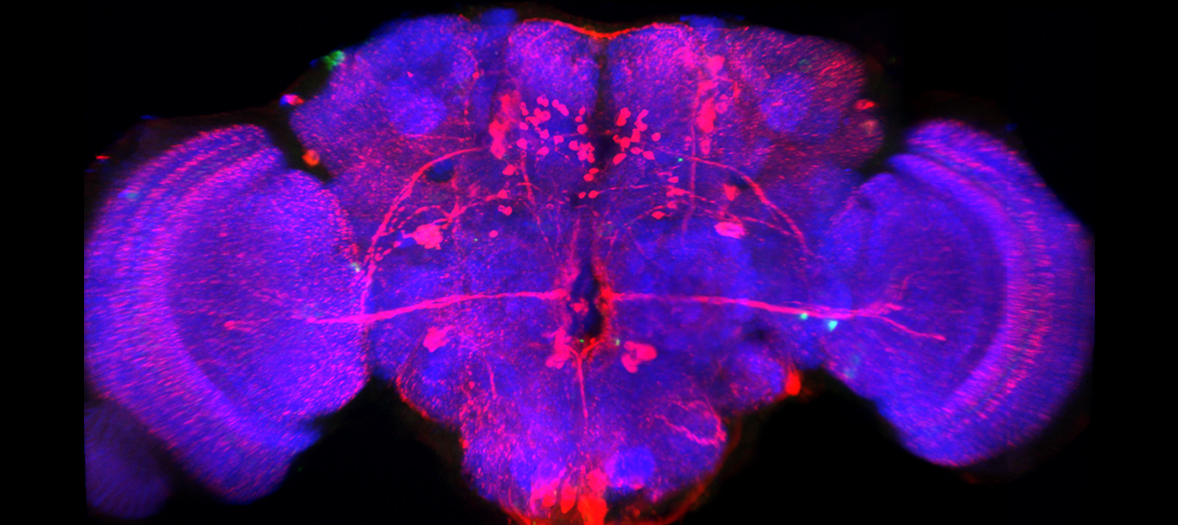
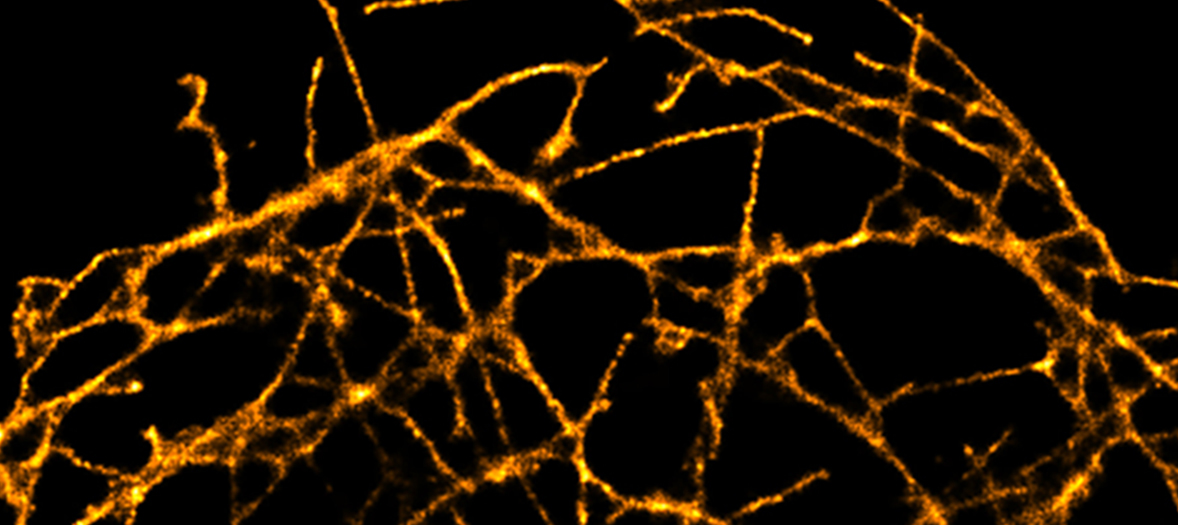
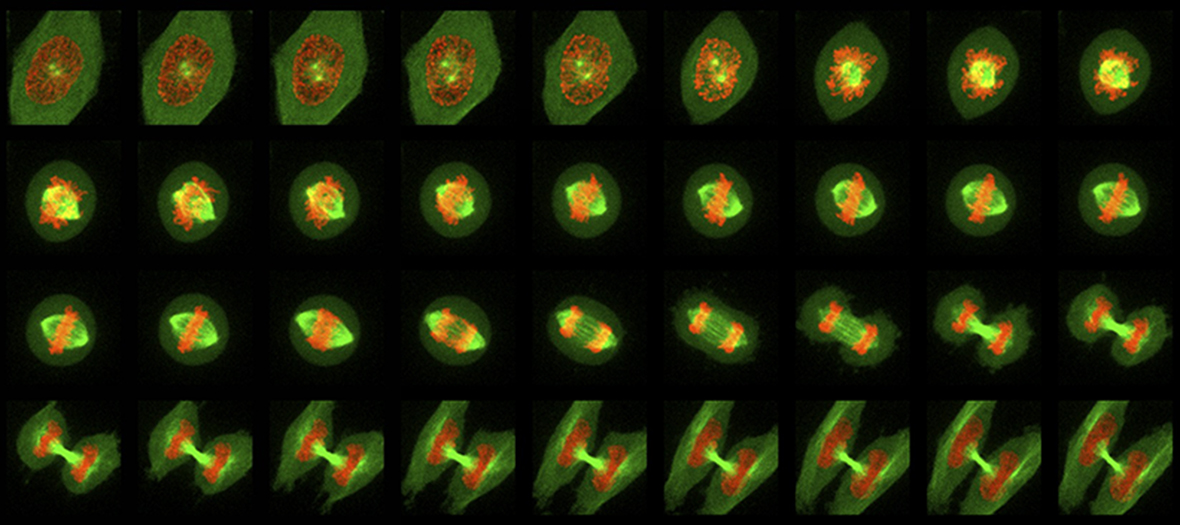
The FV1000 confocal enables imaging of thick specimens as well as fixed cells stained with standard fluorophores. Mounted on an inverted microscope, the motorized stage can be used for image stitching of large samples and imaging at multiple positions. Accompanying Imaris Software is available for 3D image reconstruction. In addition the FV1000 is fitted with a Becker and Hickl FLIM system for fluorescence lifetime imaging.
Applications:
- Imaging fixed cells and tissue sections labelled with 3 or 4 fluorophores. For investigating cellular architecture, localisation or co-localisation.
- 3D Imaging of thicker samples such as organoids, embryos etc.
- Imaging larger structures such as organ slices using image stitching.
- Imaging multiple points on a slide.
- FLIM imaging for accurate FRET interactions or assessing local fluorophore environment.
General Specifications
- FluoView Spectral FV1000 Laser Scanning Microscope based on a IX81 motorized inverted microscope
- 6 laser lines
- Freely selectable ROI for laser scanning, bleaching and photoactivation
- Scan speeds of up to 4 frames/second with 512 x 512 pixels
- xy, xz, xyz, xyzt, lambda, line scan and spot scan modes
- DIC optics
- FLIM system (Becker and Hickl) for lifetime imaging
FV1000 Laser Lines
| Laser | Excitation Lines | Suitable Dyes |
|---|---|---|
| Solid State | 405nm | Alexa 405, DAPI, Hoechst |
| Argon | 458nm | CFP |
| 488nm | Alexa 488, FITC, GFP, Cy2 | |
| 515nm | Alexa 514, YFP | |
| Solid State | 559nm | Alexa 546, TRITC, Cy3, DiI, propidium iodide, Texas red , mCherry |
| Solid State | 635nm | Alexa 633, Alexa 647, Cy5 |
Objectives Available

Selected reading:
- Cox, G. Biological confocal microscopy. Materials Today 5: 34-41 (2002).
- Fluorescence lifetime imaging--techniques and applications. Becker W. J Microsc. 247(2):119-36 (2012)
- Protein localization in living cells and tissues using FRET and FLIM. Chen Y, Mills JD, Periasamy A. Differentiation. 71(9-10):528-41 (2003).
Selected publications from the FV1000:
- Macrophages directly contribute collagen to scar formation during zebrafish heart regeneration and mouse heart repair. Filipa C. Simões, Thomas J. Cahill1, Amy Kenyon, Daria Gavriouchkina, Joaquim M. Vieira, Xin Sun, Daniela Pezzolla, Christophe Ravaud, Eva Masmanian, Michael Weinberger, Sarah Mayes, Madeleine E. Lemieux, Damien N. Barnette, Mala Gunadasa-Rohling, Ruth M. Williams, David R. Greaves, Le A. Trinh, Scott E. Fraser, Sarah L. Dallas, Robin P. Choudhury, Tatjana Sauka-Spengler & Paul R. Riley Nature Communications. 11: 600 (2020).
- ADAM17-triggered TNF signalling protects the ageing Drosophila retina from lipid droplet-mediated degeneration. Sonia Muliyil, Clémence Levet, Stefan Düsterhöft ,Iqbal Dulloo, Sally A Cowley, Matthew Freeman. EMBO J 39:e104415 (2020).
- Genetic dissection of Nodal and Bmp signalling requirements during primordial germ cell development in mouse. Senft AD, Bikoff EK, Robertson EJ, Costello I. Nature Communications. 10: 1089. (2019).
- Mitochondrial double-stranded RNA triggers antiviral signalling in humans Ashish Dhir, Somdutta Dhir, Nicholas J. Proudfoot et al., Nature. 560, 238–242 (2018).
- Intestinal Epithelial Cell Autophagy Is Required to Protect against TNF-Induced Apoptosis during Chronic Colitis in Mice. Johanna Pott, Agnieszka Martyna Kabat, Kevin Joseph Maloy. Cell Host Microbe. 23(2):191-202. (2018).
- Epithelial-mesenchymal transition transcription factors control pluripotent adult stem cell migration in vivo in planarians. Prasad Abnave, Ellen Aboukhatwa, Nobuyoshi Kosaka, James Thompson, Mark A. Hill, A. Aziz Aboobaker. Development. 144(19):3440-3453. (2017).
- Single molecule fluorescence in situ hybridisation for quantitating post-transcriptional regulation in Drosophila brains. Yang L, Titlow J, Ennis D, Smith C, Mitchell J, Young FL, Waddell S, Ish-Horowicz D, Davis I. Methods. 126:166-176. (2017).
- Phosphorylation of iRhom2 at the plasma membrane controls mammalian TACE-dependent inflammatory and growth factor signalling. Grieve AG, Xu H, Künzel U, Bambrough P, Sieber B, Freeman M. Elife. 6. pii: e23968. (2017).
- Nuclear phosphorylated Dicer processes double-stranded RNA in response to DNA damage. Burger K, Schlackow M, Potts M, Hester S, Mohammed S, Gullerova M. J Cell Biol. 216(8):2373-2389. (2017).
- Neural circuitry coordinating male copulation. Pavlou HJ, Lin AC, Neville MC, Nojima T, Diao F, Chen BE, White BH, Goodwin SF. Elife 5. pii: e20713. doi: 10.7554/eLife.20713. (2016).
- Lhx1 functions together with Otx2, Foxa2, and Ldb1 to govern anterior mesendoderm, node, and midline development. Costello I, Nowotschin S, Sun X, Mould AW, Hadjantonakis AK, Bikoff EK, Robertson EJ. Genes Dev. 29(20):2108-22. (2015).
- Cardiac lymphatics are heterogeneous in origin and respond to injury. Klotz L, Norman S, Vieira JM, Masters M, Rohling M, Dubé KN, Bollini S, Matsuzaki F, Carr CA, Riley PR. Nature. 522(7554):62-7. (2015).
Olympus FV1000

Training videos
Help Sheets
confocal microscopy