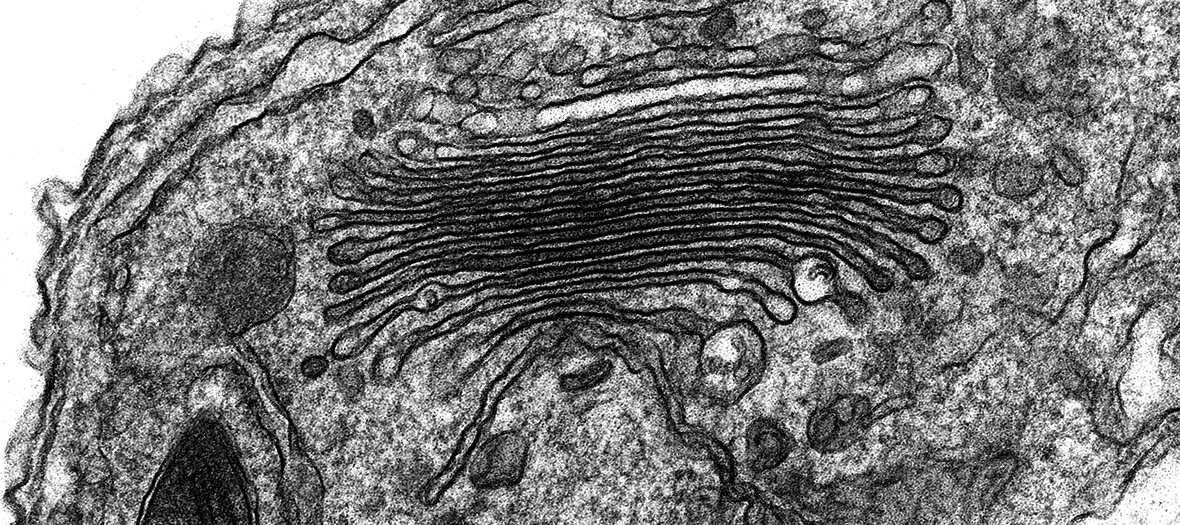
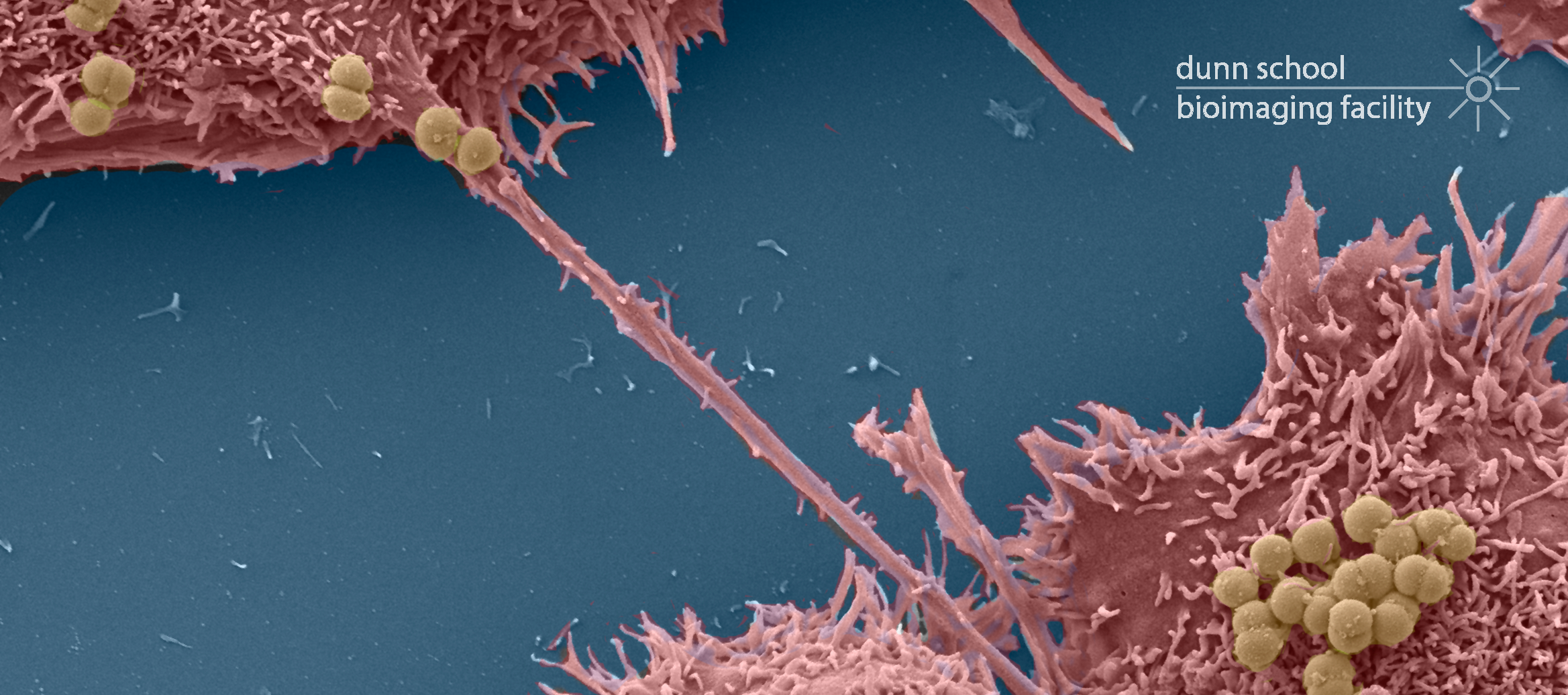
Zeiss Merlin Compact VP FEG-SEM with Gatan 3View
The Zeiss Merlin Compact Field Emission Gun SEM (FEG-SEM) with Variable Pressure (VP) mode and a Gatan 3View System was acquired in 2014 as a result of a joint BBSRC grant to Oxford Brookes University and the Sir William Dunn School of Pathology. This cutting-edge microscope contains an in situ ultramicrotome which enables the collection of large, high resolution volumes of resin embedded biological samples using a technique called serial block face scanning EM. This enables the three-dimensional architecture of whole cells and tissues to be reconstructed and analysed at the ultrastructural level.
The microscope is based at Oxford Brookes Headington campus and is jointly run by the two Departments under a shared access agreement. To learn more about the serial block face scanning EM technique and how it could benefit your research, please contact Dr Charlotte Melia: charlotte.melia@path.ox.ac.uk.
Specifications:
- GEMINI® I column with 5 pA – 20 nA Probe current
- 1 nm resolution at 15 kV
- Variable pressure mode (10-60 Pa) to mitigate charging on beam reactive samples
- In-built plasma cleaner
- Gatan 3View 2 XP system with diamond knife for 15 nm to 200 nm slices and up to 600 µm volume
- Up to 32k x 32k frame size
Applications:
- 3D ultrastructural imaging of large volumes at high resolution (eg: model the entire mitochondrial network within a cell or trace a dendrite through hundreds of microns of brain matter)
- Correlative 3D light and electron microscopy using the 3View or array tomography of serial sections
Recommended reading:
- Wilke, S.A. et al (2013). Deconstructing complexity: serial block-face electron microscopic analysis of the hippocampal mossy fiber synapse. Journal of Neuroscience, 33(2), pp.507-522.
- Kremer, A. et al (2015). Developing 3D SEM in a broad biological context. Journal of microscopy, 259(2), pp.80-96.
- Russell, M.R. et al (2017). 3D correlative light and electron microscopy of cultured cells using serial blockface scanning electron microscopy. J Cell Sci, 130(1), pp.278-291.
- Peddie, C.J. and Collinson, L.M., 2014. Exploring the third dimension: volume electron microscopy comes of age. Micron, 61, pp.9-19.
- Kay, K.R. et al (2013). Studying synapses in human brain with array tomography and electron microscopy. Nature protocols, 8(7), pp.1366-1380.
- Belevich, I. et al (2016). Microscopy image browser: a platform for segmentation and analysis of multidimensional datasets. PLoS Biol, 14(1), p.e1002340.
Publications from the Dunn School EM Facility (SBF-SEM):
- Peterzan, M.A., Clarke, W.T., Lygate, C.A., Lake, H.A., Lau, J.Y., Miller, J.J., Johnson, E., Rayner, J.J., Hundertmark, M.J., Sayeed, R. and Petrou, M., 2020. Cardiac Energetics in Patients with Aortic Stenosis and Preserved versus Reduced Ejection Fraction. Circulation. 141:1971–1985.
- Roque, H., Saurya, S., Pratt, M.B., Johnson, E. and Raff, J.W., 2018. Drosophila PLP assembles pericentriolar clouds that promote centriole stability, cohesion and MT nucleation. PLoS genetics, 14(2), p.e1007198.
- Sanchez, M.A., Tran, K.D., Valli, J., Hobbs, S., Johnson, E., Gluenz, E. and Landfear, S.M., 2016. KHARON Is an Essential Cytoskeletal Protein Involved in the Trafficking of Flagellar Membrane Proteins and Cell Division in African Trypanosomes. Journal of Biological Chemistry, 291(38), pp.19760-19773
Resources
SBF-SEM
SBF-SEM